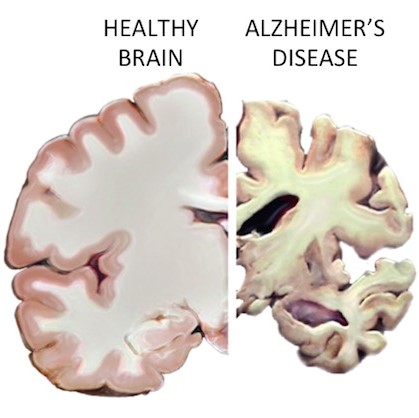
Helping disseminate information on Alzheimer’s.
For 5 years Cygnus operated the Alzheimer’s Disease Education and Referral (ADEAR) Center, for the National Institute on Aging (NIA). The primary goal of the Center was to provide information on Alzheimer's disease, its consequences and potential treatments, new research, and available services to physicians and other health professionals, patients and their families, and the public through a 1-800 toll-free telephone service. Specific ADEAR Center tasks included the following:
-
Developing and implementing systems to respond to written, e-mail, walk-in, telephone, and fax queries with information and publications;
-
Responding to queries from health professionals, patients and their families, and the lay public;
-
Preparing and disseminating written materials on the nature of the disease, its symptoms, and appropriate methods of caring for and supporting patients and family members;
-
Identifying, collecting, analyzing, storing, and retrieving materials for the Alzheimer’s disease subfile of the Combined Health Information Database (CHID) and the Alzheimer’s Disease Clinical Trials Database;
-
Designing and developing educational and informational materials, including bibliographies, bulletins, directories, factsheets, pamphlets, reports, newsletters;
-
Providing graphic design services, production, web design and development of a web site for the general public and other stakeholders;
-
Translating scientific and technical research information into information that is clearly understood by the public;
-
Developing and coordinating networks and contacts with intermediary organizations to help the ADEAR Center accomplish its mission;
-
Defining and implementing a means and media to inform and guide health professionals and reach out to the general public and minorities; and
-
Supporting the ADEAR Center at both on-site and off-site scientific and administrative meetings.